MICROSOFT SOURCE
Tiny but mighty: The Phi-3 small language models with big potential (opens in new tab)
April 23, 2024

Microsoft Research Blog
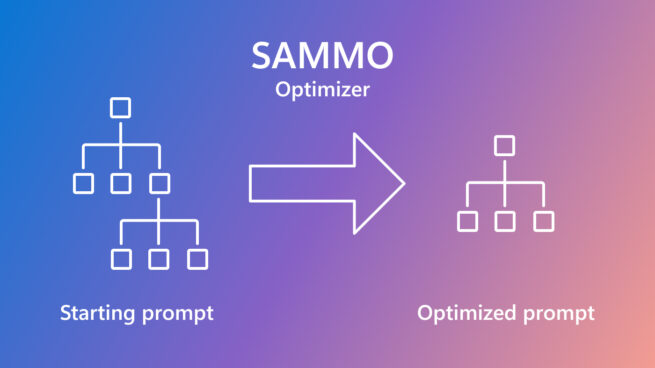
SAMMO: A general-purpose framework for prompt optimization
April 18, 2024 | Tobias Schnabel, Jennifer Neville
Microsoft Research Blog
Microsoft at NSDI 2024: Discoveries and implementations in networked systems
April 16, 2024 | Ranveer Chandra